oooo
oooo
oooo
oooo
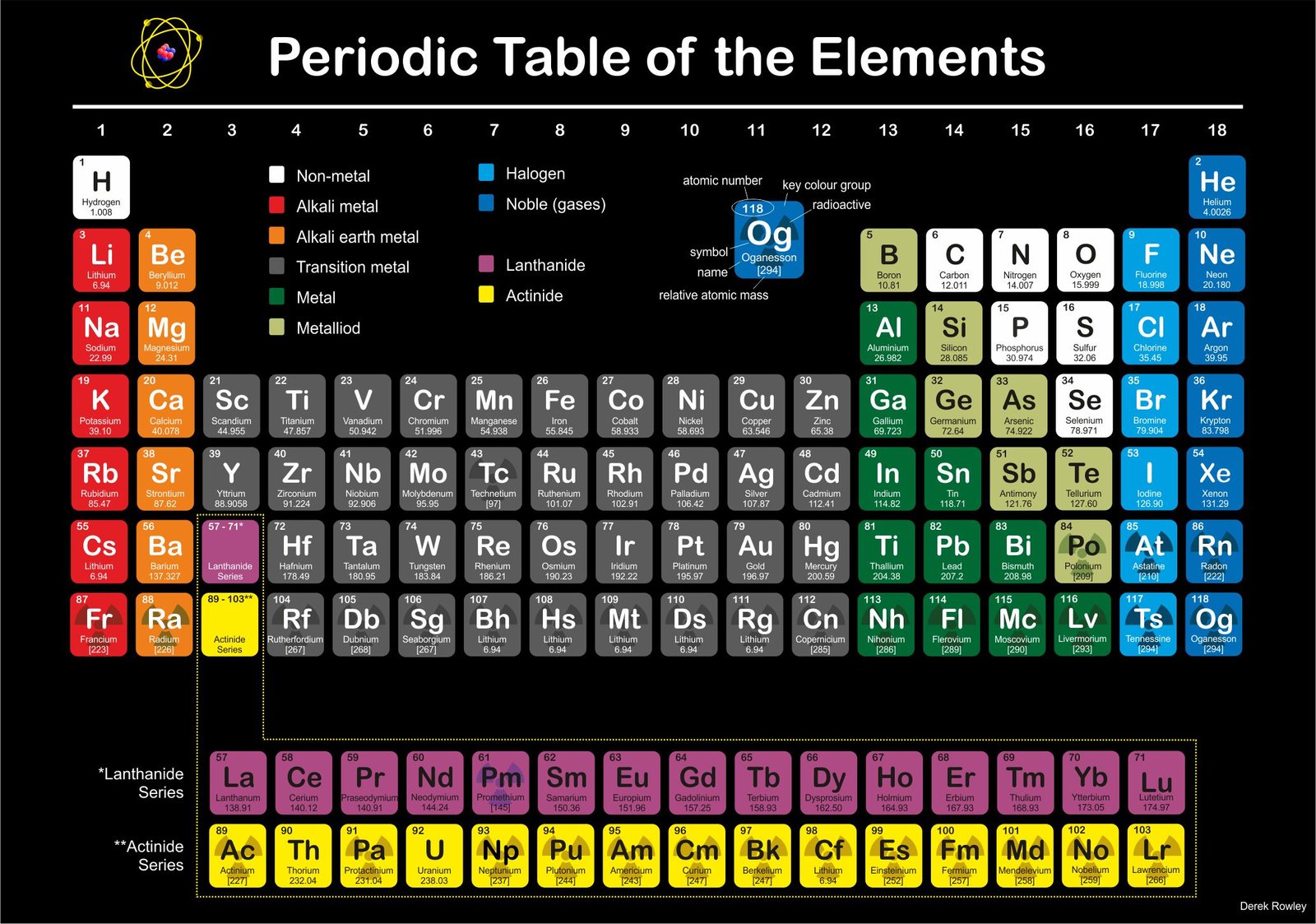
A pictorial periodic table of elements
oooo
- All the atoms that make up a single element have the same number of protons.
o
o - All atoms except those of the simplest form of hydrogen also contain neutrons.
o
o - Electrons circle the nucleus at different distances depending on how much energy they have.
o

BSL Version
oooo
oooo
Elements
oooo

- photons – that either travels as waves or as tiny particles called photons.
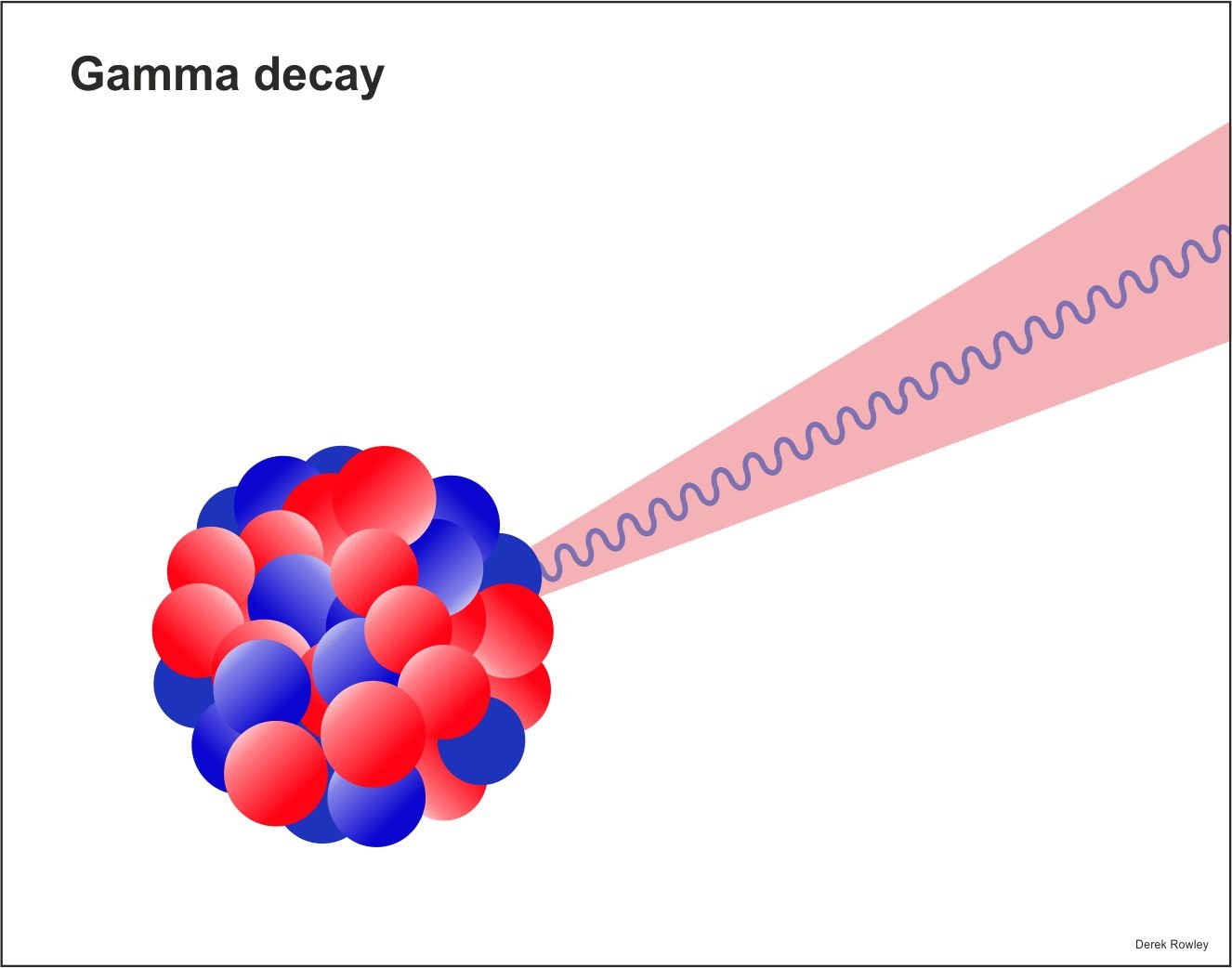
- gamma rays radioactivity – atoms decays (break down) to send out gamma rays and particles.
oooo
oooo
oooo

BSL Version
oooo
oooo
Electromagnetic wavelengths
oooo
oooo
oooo
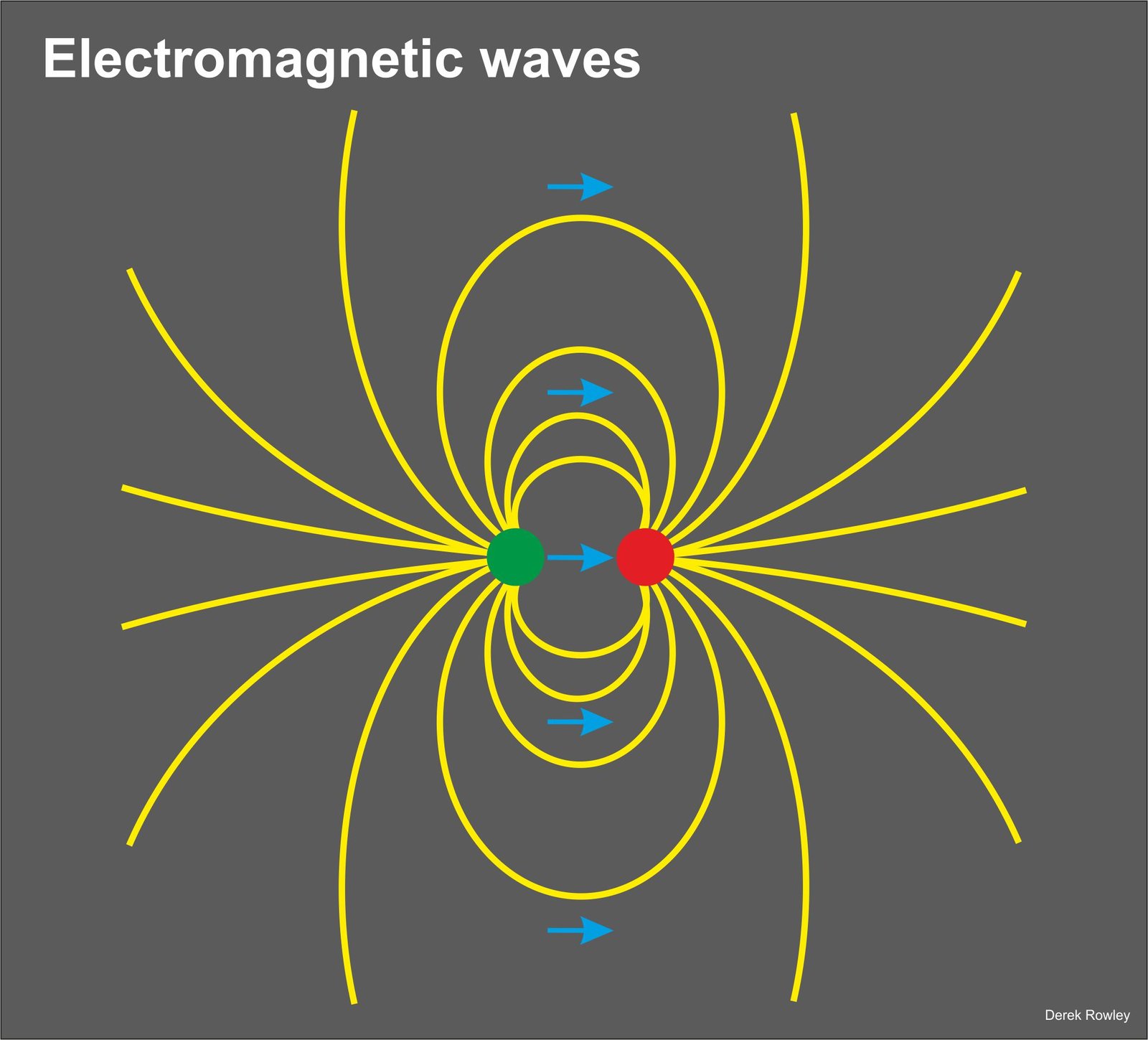
- electromagnetic radiation is electric and magnetic fields that move in tiny bursts of waves or photons.
o

BSL Version
oooo
oooo
Type of Electromagnetic radiation
oooo
oooo

oooo
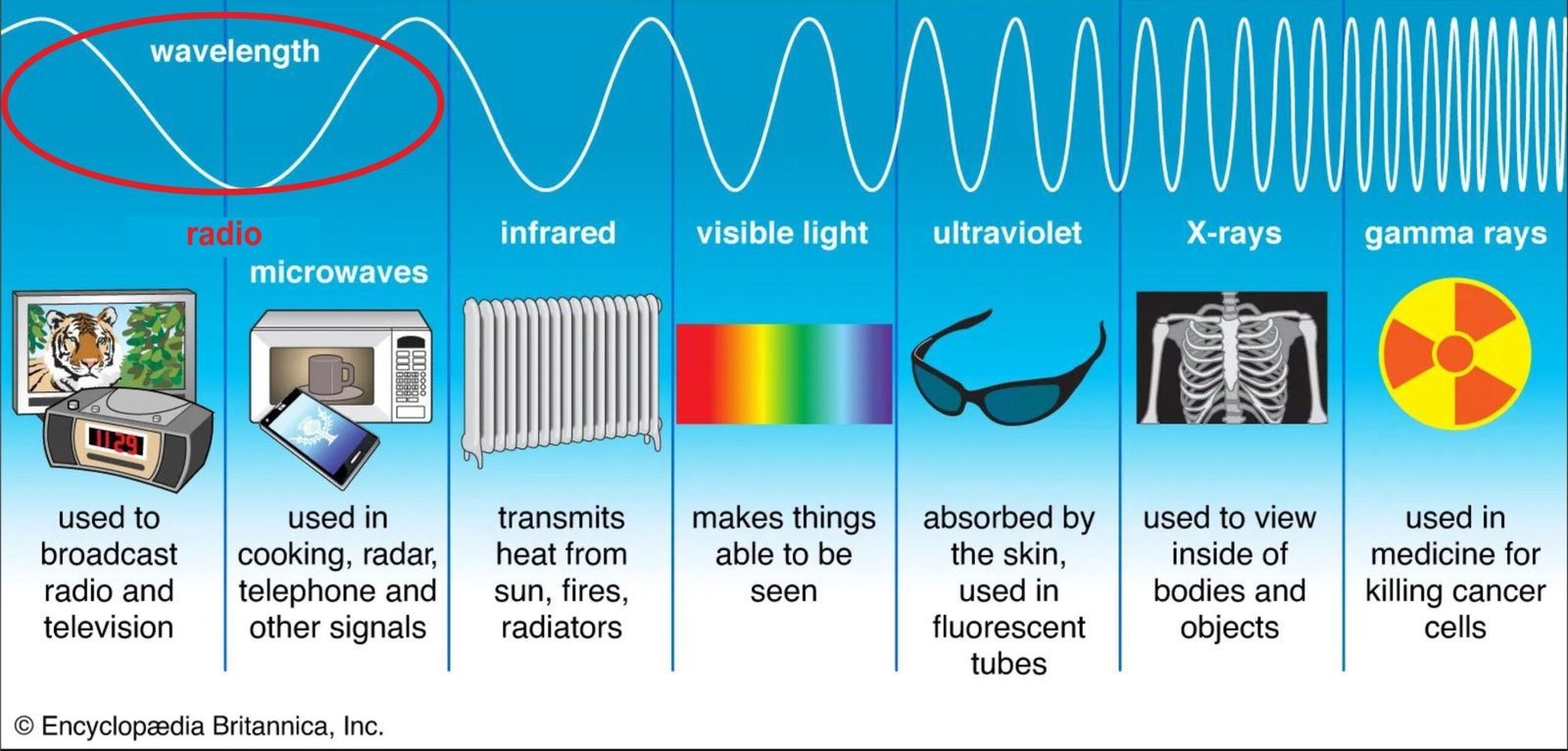
- there are different kinds of electromagnetic radiation, each with different wavelengths.
o - together, these forms of electromagnetic radiation are called the electromagnetic spectrum.
o - all electromagnetic rays move the speed of light – 299,792,458 metres per seconds.
o - everything that can be detached in space is picked up by the radiation that it gives out.
- radio waves, infrared rays, visible light, ultraviolet rays, X-rays, and gamma rays are all types of electromagnetic radiation.
o - radio waves have the longest wavelength.
o - gamma rays have the shortest wavelength.
o
o

BSL Version
oooo
oooo
Gamma Rays
oooo
oooo
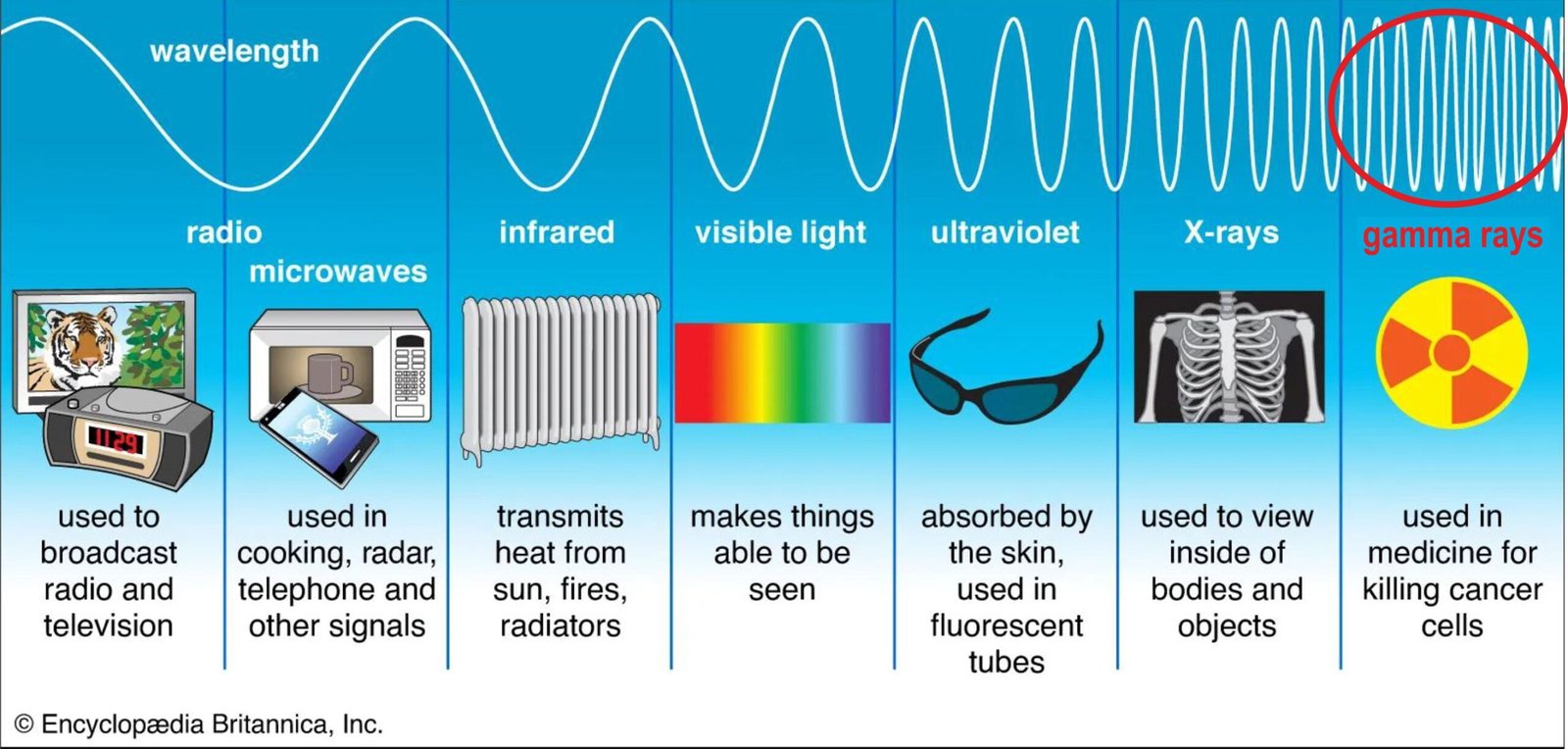
- very short wave
o - energetic and dangerous form
of electromagnetic radiation.
o - gamma rays telescope discovers extremely luminous gamma-ray sources (see YouTube).
o
oooo
oooo

BSL Version
oooo
oooo
oooo
oooo
oooo
Back to The Universe / next to Nuclear Energy page.